 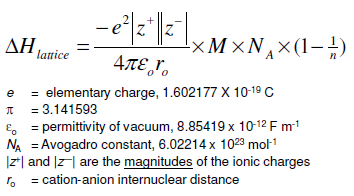
There are several different equations, of various degrees of complication, for calculating lattice energy in this way. If you know how to do it, you can then fairly easily convert between the two. The Born-Landé equation was derived from these two. 
Going across the periodic table, atomic radii decreases, therefore lattice energy increases. Calculations of this sort end up with values of lattice energy, and not lattice enthalpy. To solve for the Born-Landé equation, you must have a basic understanding of lattice energy: Lattice energy decreases as you go down a group (as atomic radii goes up, lattice energy goes down). By doing physics-style calculations, it is possible to calculate a theoretical value for what you would expect the lattice energy to be. Let's also assume that the ions are point charges - in other words that the charge is concentrated at the center of the ion. Let's assume that a compound is fully ionic. Theoretical Estimates of Lattice Energies Once again, the cycle sorts out the sign of the lattice enthalpy. This time both routes would start from the elements in their standard states, and finish at the gaseous ions. You cannot use the original one, because that would go against the flow of the lattice enthalpy arrow. It does, of course, mean that you have to find two new routes. The only difference in the diagram is the direction the lattice enthalpy arrow is pointing. How would this be different if you had drawn a lattice dissociation enthalpy in your diagram? Your diagram would now look like this: So, from the cycle we get the calculations directly underneath it. The diagram is set up to provide two different routes between the thick lines. Now we can use Hess' Law and find two different routes around the diagram which we can equate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
